In this article, we will have to discuss the properties of fluids which have a very significant role. At the start of this article, we need to know about some basic terminologies.
Table of Contents
Fluid and Fluid Mechanics
- The substances which have the ability to flow are known as Fluids. They can be liquids or gases.
- Fluid can be flow continuously as a result of a shearing action.
- It is a branch of applied mechanics that deals with the statics and dynamics of fluids.
- The study related to fluid in rest or stationary is called static fluids otherwise it is known as dynamic fluids.
Properties of Fluid Flow
Here are some of the specific properties of fluid flow:
1)Density of Fluid
The Density of fluid is the property that deals with the mass and volume of the fluid. The Density of a fluid is the ratio of its mass to the volume of that fluid.
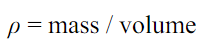
Its formula can be expressed as:
- It is denoted as: ρ (rho)
- S. I Units = kg/m3
- Its Dimension is ML-3
- This table shows the typical values of density:
| Water | 1000 kg/m3 |
| Mercury | 13546 kg/m3 |
| Air | 1.23 kg/m3 |
| Paraffin | 800 kg/m3 |
2)Specific Weight
The Specific weight/density of a fluid is the weight per unit volume of fluid. It is also called as weight density of a fluid. Its formula can be expressed as:

- It is denoted as γ
- S. I Units =N/m3
- Its Dimension for specific weight is ML-2 T-2
- This table shows the typical values of specific weight:
| Water | 9814 N/m3 |
| Mercury | 132943 N/m3 |
| Air | 12.07 N/m3 |
| Paraffin | 7851 N/m3 |
3)Relative Density (Specific Gravity)
This property of fluid can be defined as the ratio of the mass density of the fluid to the mass density of the standard fluid. In the case of liquids, the standard fluid is Water while in the case of gases the standard fluid is taken Air.
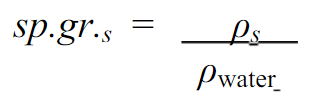
- S. I Units: none as it is a ratio
- Dimension: 1
- This table shows the typical values of specific gravity:
| Water | 1 |
| Mercury | 13.5 |
| Air | 0.00123 |
| Paraffin | 0.8 |
4)Viscosity
Viscosity is a measure of resistance to fluid flow as a result of intermolecular cohesion.
It is the property of the fluid that offers resistance to the movement of one layer of fluid over the other adjacent layer of fluid. We can also say that it is the internal resistance of a fluid to the shear and angular deformation.
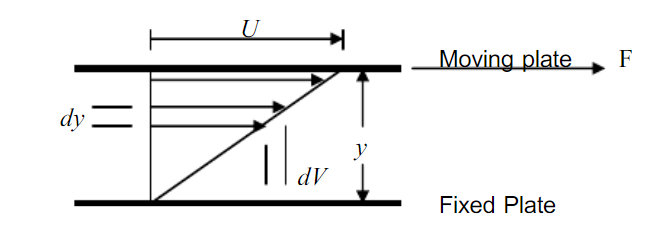
There are two ways for the expression of viscosity:
a)Coefficient of Dynamic Viscosity (µ)
The shear stress is required to drag one layer of fluid with unit velocity past another layer a unit distance away.
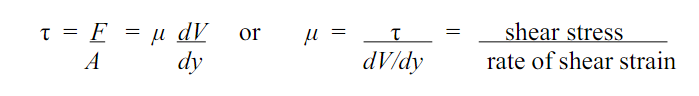
- Units for the coefficient of dynamic viscosity are Ns/m2 or kg m-1 s-1
- It is often expressed in Poise, P where (10 P = 1 Ns/m2)
- Its Dimension is M L-1 T-1
- This table shows the typical values of dynamic viscosity:
| Water | 1.14×10-3 Ns/m2 |
| Mercury | 1.552 Ns/m2 |
| Air | 1.78×10-5 Ns/m2 |
| Paraffin | 1.9 Ns/m2 |
b)Kinematic Viscosity (ʋ)
The kinematic viscosity is defined as the ratio of dynamic viscosity to density.

- Units for the coefficient of kinematic viscosity are m2/s or ft2/s
- It is usually measured in cm2/s, also called stokes
- Its Dimension is L2 T-1
- This table shows the typical values of kinematic viscosity:
| Water | 1.14×10-6 m2/s |
| Mercury | 1.145×10-4 m2/s |
| Air | 1.46×10-5 m2/s |
| Paraffin | 2.375×10-3 m2/s |
5)Weight flow rate
It is the Weight of any fluid passing through any section per unit time. Its formula is:
G = W/t = γ*V/t = γ*Q = γ*A*V
- Units for the weight flow rate are N/s or ft/s
6)Mass flow rate
It is the mass of any fluid passing through any section per unit time. Its formula is:
M = M/t = ρ*V/t = ρ*Q = ρ*A*V
7)Specific volume
It can be defined as the number of cubic meters occupied by one kilogram of any substance. It is denoted by ν.
ν = V/m = ρ-1
- Units for specific volume: m^3/kg
8)Surface Tension
When the two immiscible liquids came in contact, an unbalanced force is created at the interface stretched over the fluid mass. The intensity of force of attraction per unit length along any line of surface is known as surface tension.
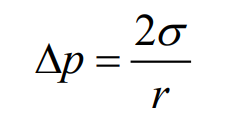
- Unit for surface tension is N/m
- Dimension for surface tension is M T-2
9)Capillarity
It is the rise and fall of a liquid in a small diameter tube that is caused by surface tension. If the diameter of the fluid is small then the combination of surface tension and adhesive forces between the liquid and container will act to propel the liquid.
10)Temperature
This is the thermodynamic property of fluids by which we can determine the state of hotness and coldness of a fluid.
- Temperature can be measured in Celsius, Kelvin, and Fahrenheit.
- Mostly Kelvin is used due to its independent from the properties of a substance
This graph clearly shows the relation between viscosity and temperature
11)Pressure (Pa)
It is one of the common properties that can be defined as force per unit area, which means that force of 1N exerts the pressure of 1 Pa (Pascal) on 1 sq. m. of area.
- Pressure can be represented as P
- S I Unit of Pressure: N/m2
- Dimension of Pressure is M L-1 T-2
12)Speed of sound
When the fluid propagates at finite velocity, it induces some disturbances in the fluid. The velocity at which these disturbances propagate is called the speed of sound.
Below is the mathematical representation of the speed of sound:

13)Vapor Pressure
It is also a property of fluids that describes the pressure at which the fluid starts to boil is called its vapor pressure. It has a direct relation with temperature, as temperature increases vapor pressure also increases.
- The Vapor pressure of water at 10˚C is 0.01atm
14) Cavitation
This property occurs when the fluid is flowing and at certain locations, the pressure is very low. That results in the formation of vapor pockets and cavities that are carried out from the origin.
15)Adhesion
It is the property of fluid that enables it to stick to another body.
16)Cohesion
The property of the liquid that resists tensile stress is called cohesion.


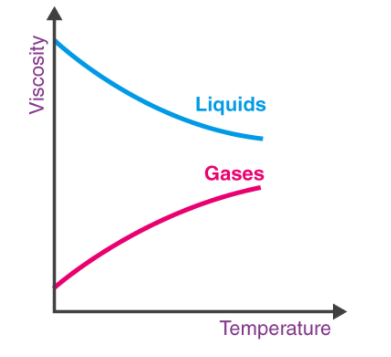 This graph clearly shows the relation between viscosity and temperature
This graph clearly shows the relation between viscosity and temperature