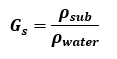Table of Contents
Standard Used
ASTM C-188
Objective
This test aims at determining the specific gravity or relative density of cement which can then be compared with the standard or reference value to hint out the possible impurities in the cement.
Related Theory
Specific Gravity:
The specific gravity (also called the relative density) of a substance is the ratio of its density to the density of water at 4°C. It is a dimensionless number and is mathematically calculated as,
The specific gravity of a substance varies with temperature.
Specific Gravity of Cement:
The specific gravity of cement is the ratio of the weight of cement to the weight of volume displaced by it. Mathematically, it can be expressed as;
The denominator in the above expression indicates the weight of the liquid displaced due to addition of cement into it.
The specific gravity of Ordinary Portland Cement (OPC) is 3.15.
For the determination of specific gravity, water is not used. This is because when water is added to powdered cement, the hydration process commences. Therefore, kerosene is used as a replacement for water.
Significance of determination of specific gravity of cement
The specific gravity of cement is an important parameter that informs about its density. In mix design procedures, this is of paramount importance since a higher value of specific gravity than that generally accepted signifies that it has more moisture content. This can possibly affect the binding property of cement, its workability, and strength.
Apparatus
- Le-Chatelier’s flask, circular in cross-section and made up of transparent glass having marked graduations
- Ordinary Portland Cement (OPC)
- Kerosene oil or naphtha, having a density greater than 0.73 g/mL at 23°C
- Glass funnel, to transfer kerosene oil into the Le-Chatelier’s flask
- Weighing balance, to weigh the required cement quantity and water
- Trowel, for handling cement
- Water, as a reference liquid for specific gravity determination
- Measuring cylinders, to measure the exact amount of water
- Water bath, to regulate and control the test temperature
Fig.: Le-Chatelier’s Flask
Test Procedure
Test Temperature
The Le-Chatelier’s flask should be immersed in a water bath sporadically to ensure the temperature variation does not exceed 0.2°C at the time of recording the readings.
- Fill the Le-Chatelier’s flask with kerosene oil or naphtha between 0 and 1mL graduations present on the flask using a glass funnel.
- Record the initial level on the flask.
- Weight about 64g of cement of a weighing balance.
- Add the weighed quantity of cement into the flask carefully in increments at the same temperature as that of the liquid present in it.
- Whirl the flask horizontally to remove air from the cement.
- After the addition of cement, the level of water in the flask will rise. Record the final reading on the flask after placing it in the water bath to prevent temperature fluctuations from affecting the test results.
- Calculate the change in the level of liquid in the flask by taking the difference of initial and final readings.
- The differential volume in the flask in actuality, is the volume of cement, the weight of which is already known (64gm). The specific gravity of cement can then be determined in two ways.
Method I
- In order to determine the specific gravity of cement, take water in a measuring flask equal to the volume of cement determined previously.
- Determine the weight of this known volume of water. This is termed as the weight of an equal volume of water, or the weight of displaced volume.
- Calculate the specific gravity of cement as follows;
Method II
- Determine the density of cement using its known weight and displaced volume as
- Calculate the specific gravity of cement using the known water density at 4°C as
- Compare the specific gravity value with the standard specific gravity of Ordinary Portland Cement i.e., 3.15.
Observations and Calculations
Weight of cement taken (g) = Wc =
Initial flask reading (mL) = Vi =
Final flask reading (mL) = Vf =
Difference in the level of flask = Volume of cement = V = Vf – Vi
Method I
Volume of water taken in the measuring cylinder = V =
Weight of water having volume V (g) = Ww =
Specific Gravity of cement = Wc / Ww
Method II
Density of cement = ρc = Wc /V
Density of water at 4°C = ρw = 1000 g/cm3
Specific gravity of cement = ρc / ρw
Note: At least two tests must be carried out and their average value must be reported. Additionally, if the two test results differ by more than 0.03, a retest is mandatory.
Results
The specific gravity of cement is .
The standard specific gravity of cement is 3.15.
If the obtained value of specific gravity differs greatly from the standard or refernce value (given that the test is performed under standard conditions), the cement contains impurities.
Precautions
- The liquid to be added to the flask (kerosene or naphtha) should be free from water.
- The inside of Le-Chatelier’s flask should be dry before adding kerosene or naphtha into it.
- The cement that adheres to the inside of the flask should be stirred to form a heterogeneous mixture of cement and the liquid.
- Care must be exercised while adding cement to the flask to prevent it from spilling out. This is because if the weighed amount of cement does not go into the flask, the calculated results will give a misleading value of specific gravity.
- It is imperative to remove air bubbles by stirring the flask in horizontal motion. This is to prevent erroneous volumetric readings of the flask.
Discussion
- Apart from aiding in weight-volume relationships, the specific gravity of cement is an important physical parameter to assess its purity.
- Inaccurate volumetric readings of the flask and temperature conditions in disparity to the standard can be possible causes of error. For specific gravity determination, the temperature condition is quintessential to achieve, otherwise, the obtained value of specific gravity will be in deviation from the actual value.
- If the cement is kept for a longer duration and care is not exercised to prevent it from reacting with the moisture present in the atmosphere, the cement undergoes hydration. Therefore, in such situations, a retest on specific gravity determination is suggested.